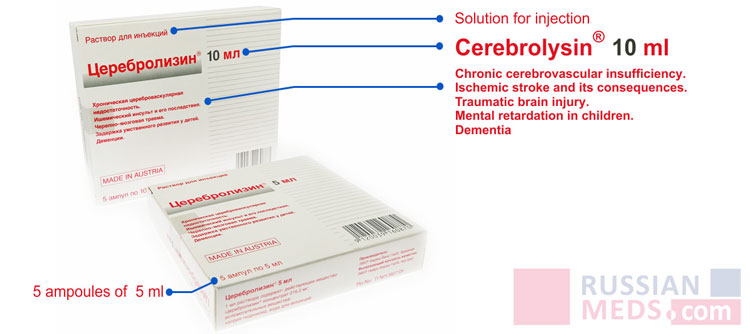
Cerebrolysin®
Cerebrolysin® is the world's bestseller for stroke, TBI, dementia or cognitive impairment patients.Cerebrolysin® improves the brain's ability to recover by stimulating the regeneration of the nervous system.
Legal Notice:
Cerebrolysin® is a registered trademark of EVER Pharma. All trademarks and brand names are the property of their respective owners. This website/store is not affiliated with, endorsed by, or sponsored by ЕVER Pharma.

Active substance:
Cerebrolysin® concentrate (pig brain-derived peptide complex)Pharmachologic effect:
nootropic, neurometabolicPharmacodynamics:
Cerebrolysin® contains low-molecular-weight biologically active neuropeptides that penetrate the BBB and directly enter the nerve cells. The drug has an organ-specific multimodal effect on the brain, i.e. provides metabolic regulation, neuroprotection, functional neuromodulation and neurotrophic activity.
Metabolic regulation: Cerebrolysin® increases the efficiency of aerobic energy metabolism in the brain, improves intracellular protein synthesis in the developing and aging brain.
Neuroprotection: Cerebrolysin® protects neurons from the damaging effect of lactic acidosis, prevents the formation of free radicals, increases survival and prevents neuronal death under conditions of hypoxia and ischemia, and reduces the damaging neurotoxic effect of excitatory amino acids (glutamate).
Neurotrophic activity: Cerebrolysin® is the only peptidergic nootropic drug with proven neurotrophic activity, similar to the action of natural neuronal growth factors (NGF), but manifested under conditions of peripheral administration.
Functional neuromodulation: Cerebrolysin® has a positive effect on cognitive impairment, improves memory processes.
Pharmacokinetics:
The complex composition of Cerebrolysin®, the active fraction of which consists of a balanced and stable mixture of biologically active oligopeptides with a total polyfunctional effect, does not allow for the usual pharmacokinetic analysis of individual components.
Indications:
Alzheimer's disease;
dementia of various origins;
chronic cerebrovascular insufficiency;
ischemic stroke;
traumatic brain and spinal cord injuries;
mental retardation in children;
attention deficit hyperactivity disorder (ADHD);
with endogenous depression resistant to antidepressants - in combination therapy.
Contraindications:
individual intolerance to the drug;
severe renal failure;
status epilepticus.
Use with caution:
allergic diathesis; diseases of an epileptic nature, incl. with generalized epilepsy (due to a possible increase in the frequency of seizures).
Pregnancy and breast-feeding:
During pregnancy and during breastfeeding, Cerebrolysin® should be used only after a careful analysis of the ratio of the positive effect of treatment and the risk associated with its implementation.
The results of experimental studies do not give grounds to believe that Cerebrolysin® has a teratogenic effect or has a toxic effect on the fetus. However, similar clinical studies have not been conducted.
Side effects:
RARELY (from 0.01% to 0.1% cases):
loss of appetite;
the alleged activation effect is accompanied by arousal, manifested by aggressive behavior, confusion, insomnia;
too fast administration of the drug may lead to dizziness;
VERY RARELY (less than 0.01% cases):
increased individual sensitivity, allergic reactions, skin reactions, pain in the neck, head and extremities, fever, mild back pain, shortness of breath, chills, collaptoid state;
isolated cases of generalized epilepsy;
too fast administration of the drug can lead to increased heart rate and arrhythmias;
slidispepsia, diarrhea, constipation, nausea, vomiting.
Since Cerebrolysin® is used mainly in elderly patients, the above symptoms of the diseases are typical for this age group and often also occur without the use of the drug. It should be noted that some undesirable effects (agitation, arterial hypertension, arterial hypotension, lethargy, tremor, depression, apathy, dizziness, headache, shortness of breath, diarrhea, nausea) were identified in clinical trials and occurred to the same extent as in patients, who received Cerebrolysin®, and in patients in the placebo group.
Interaction:
Taking into account the pharmacological profile of Cerebrolysin®, special attention should be paid to possible additive effects when administered simultaneously with antidepressants, incl. MAO inhibitors. In such cases, it is recommended to reduce the dose of the antidepressant.
The use of high doses of Cerebrolysin® (30-40 ml) in combination with high doses of MAO inhibitors can cause an increase in blood pressure.
Do not mix Cerebrolysin® and balanced amino acid solutions in the same solution for infusion.
Cerebrolysin® is incompatible with solutions that contain lipids and solutions that change the pH (5–8).
Dosing and Administration:
Intramuscularly (up to 5 ml),
intravenously (up to 10 ml),
intravenously by slow infusion (10 to 50 ml).
Doses and duration of treatment depend on the nature and severity of the disease, as well as on the patient's age. Perhaps the appointment of single doses, the value of which can reach 50 ml, but it is more preferable to carry out a course of treatment. The recommended optimal course of treatment is daily injections for 10–20 days.
Acute conditions (ischemic stroke, TBI, complications after neurosurgical operations) - from 10 to 50 ml.
The residual period of cerebral stroke and traumatic injury to the brain and spinal cord is from 5 to 50 ml.
Psychoorganic syndrome and depression - from 5 to 30 ml.
Alzheimer's disease, dementia of vascular and combined Alzheimer's-vascular genesis - from 5 to 30 ml.
In neuropediatric practice - 0.1–0.2 ml / kg.
To increase the effectiveness of treatment, repeated courses can be carried out until the patient's condition improves as a result of treatment. After the first course, the frequency of prescribing doses can be reduced to 2 or 3 times a week.
Doses from 10 to 50 ml are recommended to be administered only by slow intravenous infusions after dilution with the proposed standard solutions for infusion. The duration of the infusion is 15 to 60 minutes.
Overdose:
No cases have been identified.
Special instructions:
If injections are performed too quickly, a feeling of heat, sweating, dizziness may occur, so the drug should be injected slowly.
The compatibility of the drug (within 24 hours at room temperature and in the presence of light) with the following standard solutions for infusion has been tested and confirmed:
- 0.9% sodium chloride solution (9 mg NaCl / ml);
- Ringer's solution (Na + - 153.98 mmol / l; Ca2 + - 2.74 mmol / l; K + - 4.02 mmol / l; Cl− - 163.48 mmol / l);
- 5% glucose solution.
Simultaneous administration of Cerebrolysin® with vitamins and drugs that improve cardiac circulation is allowed, however, these drugs should not be mixed in the same syringe with the drug Cerebrolysin®.
Use only a clear solution and only once.
Clinical trials have shown that Cerebrolysin® does not affect the ability to drive vehicles and use mechanisms.
Storage:
The temperature is not above 25 °C (77 °F). Do not freeze.
Cerebrolysin®
in Cyrillic : Церебролизин
SUBSTANCE
Cerebrolysin
DOSAGE FORM
ampoules
MANUFACTURER
In Stock
available pack options :
| 2ml x 10 ampoules | $38.90 | |
| 5ml x 5 ampoules | $39.40 | |
| 10ml x 5 ampoules | $55.90 |